Backyard Hydrogen Generator: Recap
For those who don't want to wade through 4 posts of stream-of-consciousness
Q: What is this, what is it called, and what can it do?
A: This is a backyard hydrogen generator that can produce a few kilos of hydrogen in an hour or two using methane (from a natural gas line) or propane (from a tank at Wal-Mart) as a feedstock, using electricity and water. The tentative name is Pythia.
Q: What would I use it for and where do I buy one?
A: You could use it to fill weather balloons or do backyard chemistry. I’m sure there are other things it would come in handy for, but we need tinkerers playing with it. It’s not for sale yet because I’m trying to get the capital together to build a prototype and start manufacturing them. I am hoping Kickstarter will approve a post on it.
Q: How does it work?
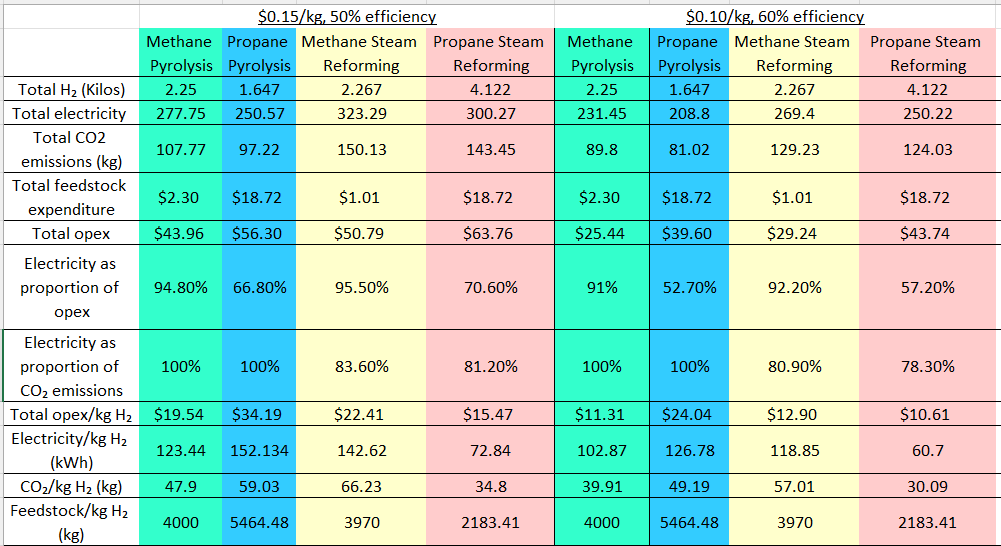
A: Either pyrolysis, which heats the feedstock directly to produce hydrogen and carbon soot, or steam reforming, which reacts it with steam. Depends on your situation. See this chart (note: some numbers are off, but in the sense that they underestimate rather than overestimate hydrogen production:
In general, methane pyrolysis is a good choice if you want to minimize carbon emissions. It requires higher temperatures and may take some more finagling to get working. Propane pyrolysis will only be a good choice in a few situations. Steam reforming is well understood chemically but requires an extra step or two.
Q: Is it dangerous?
Shouldn’t be if you’re not an idiot. Don’t smoke around this.
Q: How does it work?
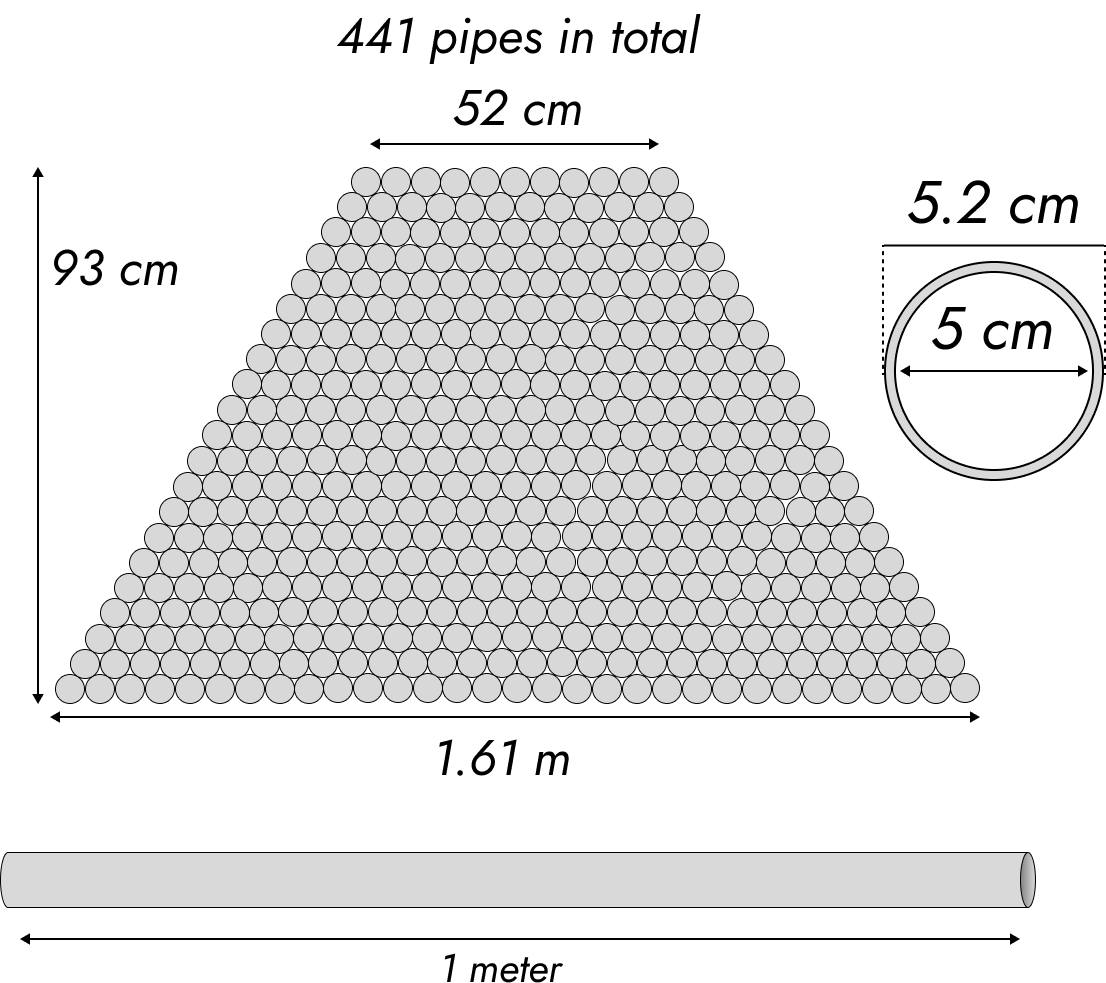
Your feedstock (that’s the methane or propane) and, in the case of steam reforming, steam gets fed into hundreds of little tubes made out of either iron or a nickel alloy via a smaller tube inside it. Inside that even smaller tube is a rod of silicon carbide, which heats it up to really high temperatures, around 900-1000C. Here’s a cross-section.
Hydrogen comes out the other end, along with CO₂ in the case of steam reforming.
Q: Will this explode?
A: No. The pressures involved are quite low and there will be safety valves.
Q: How do you separate the CO₂?
Well, technically, what’ll come out the other end is a mixture of hydrogen, CO₂, water and carbon monoxide (CO), so we’ll send that to a second chamber to undergo the water-gas shift reaction at around 350-400C, where the CO will react with steam to produce CO₂ and another hydrogen.
Then we’ll send it off to be bubbled into a tank of limewater, which will absorb the CO₂. H₂ will collect at the top of the tank, where you can draw it off.
Q: How do you store it?
A: You don’t, really, this is produce-on-demand. A weather balloon should keep some around for a day or so, but it’ll leak. Hydrogen’s tiny.
Q: Do you need catalysts?
A: Nickel for steam reforming and propane pyrolysis, and iron for methane pyrolysis, which is what the pipes are made of. Ideally, Pythia will be able to do either steam reforming or pyrolysis as you prefer, but the correct choice is usually pretty specific to the end user and we may end up doing two different models.
Q: How big is this thing?
A: The tubing system should be probably about 1.6m wide by a meter high by a meter long. At least, that’s a good ballpark figure.
The gas-shift reactor should be smaller than that. The limewater reactor is probably going to be rather large because hydrogen takes up so much room (4 kilos at 100 psi and room temperature is about 7 cubic meters; you’ll probably need to draw it off as it’s produced.)
All the components should be modular, so you can bury the limewater reactor, e.g., in your backyard, where you’re likely to have more space.
Q: Those are very high temperatures.
A: The metals in question are pretty sturdy, and we’ll design it carefully so that heat isn’t lost unnecessarily. For example, the steam pipes for the water-gas shifter are probably going to be snaking around the main reactor to draw off waste heat and make use of it in the steam reformer, which doesn’t need to run as hot. And we’re heating what are, in the grand scheme of things, relatively small amounts of gas, so with good insulation and energy efficiency it should be fairly affordable.
The big expense is electricity, so if you have your own solar set or live in an area where you can run appliances off-peak for pennies, you should do that.
Q: What is the projected capex cost?
A: We are not sure yet, but none of the components are inherently pricey. The necessary tubing should be less than a grand for raw material, though the seals with the intake valves need to be good. Silicon carbide is decently cheap.
Q: How much hydrogen can this produce?
A: We are going to shoot for 4 kilos an hour. That’s definitely possible for pyrolysis and should be feasible for steam reforming. Again, you’ll need to draw it off as it’s produced.
Q: What happens if oxygen gets in?
A: Throw away the pipe (they’ll be modular, remember), then flush truly teeny amounts of propane into other components as a scavenger. We’ll be using one-way valves to ensure that gases only go the direction they’re supposed to be going in.
Q: Is this legal?
A: As far as I know, yes—you can buy hydrogen generators on Amazon, only they run on electrolysis and are weaksauce.
Is it legal for you to run it? Check local laws. We are going to be getting legal counsel re: product liability.
Q: What is needed now?
Become a paid subscriber to subsidize legal costs and production, email me at campbell.nilsen@gmail.com, or DM me on Twitter at @nephew_jonathan. Once I’ve cleared the legal hurdles, early experimentation shouldn’t be that expensive; if we can get hydrogen out of a single pipe, we can get it out of 500.
I live in the DC area, so I’ll be looking for a semi-abandoned barn or house in, probably, the West Virginia Panhandle to turn into a lab. A property with a gas line hookup is ideal.