Backyard Hydrogen Generator: Update 1
In which we learn some more materials science and eventually cut our costs by a factor of, like, five
(Continued from here.)
I am happy to report that I’ve gotten my first ballpark quote for a high-temperature, high-pressure reaction chamber at 900C with a copper lining: about $15K, from the wonderful folks at Alloy Engineering. About $9000 of that is the materials. This was for a large-ish vessel with a height of about a meter and a diameter of 20cm…though I did say “two vessels” at one point with an eye towards the water-gas shifter so it might be more like $4500 in materials. (The water-gas shifter won’t be getting anywhere near as hot, though.)
Most steels and copper aren’t going to do crazy well with repeated trips up to 900C, so they used a nickel-chromium alloy for the quote, which drove the price up considerably. This raises the question of whether copper is necessarily the right choice. Its permeability is low, but this is also true of a number of ceramic materials. Let’s look at those.
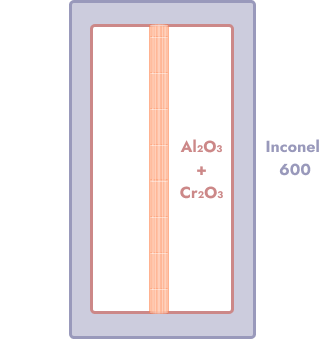
This paper gives a pretty good overview of the hydrogen permeability of various potential coatings. Good options appear to include aluminum oxide, chromium oxide1, and a mixture of the two. Both Al₂O₃ and Cr₂O₃ have very high melting points and don’t risk deformation in the 900C range.
Major open questions on the material front now boil down to:
a) the right heating element
b) the correct structural alloy
For a), I’m going to assume Nichrome wire. Note that we really don’t want any oxygen in the steam reformer while the thing is operating. Getting a decent vacuum in the thing before you start running it and doing a small dry run with some propane before you really start cycling should take care of that, as the propane will act as a scavenger for spare oxygen (autoignition temperature is 450C, though that’s at ambient pressures and oxygen concentrations). Scavenging for spare hydrogen in between runs is probably not a bad idea, but again, a good pressure gauge should be able to tell us whether the pressure inside the reactor could possibly be enough to be risky (we’d want to assume, for safety, that all the pressure is caused by hydrogen).
Now the structural alloy.
(Note: while working all this out it eventually became apparent that metal isn’t the correct structural option. Skip down here to continue with discussion of quartz and ceramics.)
Nichrome wire running down the length of the tube is probably fine, in fact, because under normal circumstances (i.e. with oxygen around) Nichrome develops a protective coating of Cr₂O₃ that protects it from further oxidation and, as a bonus, is pretty impermeable to hydrogen. (So we’ll have to do an early run in the presence of air to get the protective coating on the nichrome before we start running things through.
We could try something like tubing made of Inconel, whose solidus temperature (i.e. the temperature below which it is absolutely, no questions asked, a solid) is in the 1300C range—well above what we need. Inconel 600 appears to be in the $30-40/kg range.
This data sheet gives us a wealth of info about the properties of inconel, which combined with this handbook of pressure-vessel math (no more thumbing through pirated homework cheat sheets!) can give us a good idea of how much material we’re looking at.
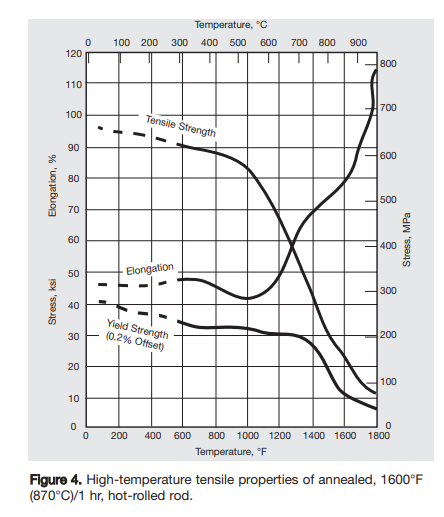
Shear strain isn’t relevant for pressure vessels, at least not to a first approximation, so let’s take a good look at tensile strength, since (almost) all stresses in a pressure vessel will be tensile. Tensile strength starts to collapse in relative terms for inconel at high temperatures:
There are two main models for stress on a pressure vessel, depending on whether it’s thin-walled or thick-walled. It’s “thin-walled” if the inner diameter is more than 20 times the wall thickness, so we’ll look at that first (both because we’d like to save on material and because the math’s much, much easier).
We’ll only look at hoop stress (σ_θ2) for now, since the top and bottom halves of the pressure vessel are more affected by longitudinal stress, σ_L, which is only half the hoop stress given constant wall thicknesses throughout the vessel. (There’s also radial stress, σ_r, which is negligible for thin-walled vessels).
For a cylinder or cylinder-like3 vessel with inner diameter d and wall thickness t, then σ_θ at a given pressure p is given by our old friend (p·d)/(2t)4; a more helpful way of thinking about this is that (2σ_θ / p) = (d/t), or in other words that twice the ratio of the strain to the operating pressure is going to be the same as the ratio of the diameter to the wall thickness.
Hoop stress is tensile stress; tensile strength at 900C per the graph is somewhere around ~15,000 psi (15 ksi), but we’d like to be cautious, so we’ll pretend it’s a third of that. Then (10,000/p) = (d/t); at 200 psi, the diameter needs to be no more than fifty times wider than the wall thickness, which seems pretty good; let’s say forty times wider for extra safety5.
We’ve now got an optimization problem of sorts. Doubling the diameter at a contant pressure also entails doubling the wall thickness, which a little bit more than doubles the material used, but also quadruples the volume. Alternatively, we can halve both the pressure and the wall thickness. This also answers a conundrum I was considering for a short period as to whether it might not be more efficient to have multiple small reactor vessel; the answer is no, because volume of material you can react in a single go quadruples every time you just-about-double the structural alloy required by doubling the wall thickness.
To translate this into capex terms, let’s assume we want a pressure vessel that’s 0.5m³ in volume. If it’s shaped like a cylinder with length h and inner radius r capped by hemispheres at both ends, then V = hπr² + (4πr³)/3. Optimal volume-to-surface-area ratio can be found if you have a sphere and h is zero, but spheres are inconvenient to move around, so we’ll go for h = 2r. Then 500,000cm³ = (10πr³) / 3, r is about 36.2cm and both d and h are about 72.5cm. Wall thickness then needs to be around 1.8cm.
The total volume of wall material, if I’ve gotten my math correct here, should be somewhere around 63,100 cm³—let’s call it 64,000 cm³ just to be safe. Inconel’s density is 8.47g/cm³, so that’s 542 kilos, or a price of about…ah, cripes, $16-20K at prices of around $30-40 a kilo—
That price is insane.
Yeah, screw it, let’s look at other options.
An alloy is considered high-temperature-grade if it won’t fall apart in the 8-900C range, but for ceramics the line starts at ~2000C, well above what we need or want. Fused quartz tubes will operate up to 1200C, though they start doing funny things around 1100C, and they have very good thermal shock properties. They’re brittle, of course (this is glass!), but sufficiently thick quartz tubing should work pretty well, and it’s cheaper than metal.
The main question is the tensile strength involved: this old paper (figure 10) found a mostly linear decline in the tensile strength of fused quartz fibers as temperature rose. They only went up to 800C, and there’s some sort of unit weirdness going on when I try to convert kilogram-force per mm² to psi—one kg/mm² is supposed to be about 1425 psi, which means the tensile force at 900C, if we’re eyeballing it, should be north of 100,000 psi—which can’t be right; fused quartz seems to have a usual tensile strength of around 7,000 PSI.
It’s quite clear that the tensile strength of quartz is heavily dependent on manufacturing, so let’s…you know, let’s leave the calculations for a later date. Unlike fancy metal alloys, we have a lot of room here.
Quartz tubes are cheap. It’s hard to get a sense of how cheap, exactly; labware made in the US is marked up because it’s bought with grant money. This Alibaba outfit sells pretty large (diameter in the 60cm range) quartz glass tubes with a thickness of up to a centimeter for a few bucks. Unlike fancy alloys, we can easily and cheaply compensate for uncertainties about tensile strength by slapping another couple centimeters on the wall thickness. Fused quartz also has one of the strongest resistances of any substance to thermal shock, and its coefficient of thermal expansion is practically zero. Vacuum-deposit some hydrogen-proof ceramics on the inner surface and…
…we should be good to go?6
Continue with update 2.
Specifically chromium (III) oxide. No worries about hexavalent chromium here.
No subscript version of θ or capital L in Unicode.
Hemispheres are stronger than flat drum surfaces, so that’s likely to be the structure used, but flat drum surfaces are easier to make diagrams of, since the cross-section of the vessel is a rectangle.
The length of the vessel L appears in both the numerator and the denominator and thus cancels out to a first approximation. It reappears when you start looking at how much the dimensions of the vessel change, which we’ll get to in a second.
We might need to be even more cautious than this, though. We’ll find out—hopefully from an expert more than from empirical data.
We’re still gonna need some high-temp alloys for the tubing, I think.




One assumption you are making is that your strength is at temperature. You could have a setup consisting of something strong, and cheap, at ambient temperature. Followed by something thermally insulating and under compression. Followed perhaps by an inner hydrogen resistant skin.