Let’s say you—an individual customer—would like to buy some hydrogen. (There might be any number of perfectly responsible reasons why you need some). At wholesale, industrial prices, hydrogen is fairly cheap: just $2 a kilo for grey hydrogen made by steam reforming of methane, with attendant CO₂ emissions.
But you can’t get retail hydrogen at anything like that price. This isn’t so much because it’s flammable (there are all sorts of dangerously flammable things you can buy at the hardware store, of course), but because hydrogen molecules, H₂, are small and difficult to pressurize.
I’ll just get an electrolysis machine, then. That’ll be cleaner, too, no carbon emissions. You mean like one of these?
423 cubic feet, or one kilo, of H₂ is a hair under 12 cubic meters (11.978 m³, to be precise). One cubic meter is a thousand liters, so you’ll need to run this baby for about 24,000 minutes straight, or about 16 days and 16 hours (400 hours). That’ll run you 72 kilowatt-hours. At prevailing electricity prices and carbon intensities in the US ($0.15 and 389 grams of CO₂ for every kilowatt-hour), you’ll spend about $18 and be responsible for about 28 kilos of CO₂. (Less in California or Washington state, of course). And did you have somewhere to put that H₂ where it wasn’t going to leak out?
This post is an early white paper for a reasonably cheap (<$1000 with a bit of manufacturing scale), fast, highish-throughput (a few kilos an hour) backyard hydrogen generator. It runs on nothing more complex than propane, water, electricity, and chalk.
Its carbon footprint, given prevailing electricity intensities, should be lower than that of a 500mL/min generator off Amazon. It should be relatively safe if you aren’t a moron. While I wait for Kickstarter to decide that this is something you can back, here are the general details. This will be a fairly quick overview because I’ve already Tweeted myself nearly to exhaustion, so not everything will get a proper literature citation.
Industrial hydrogen is usually made via steam reformation with methane (natural gas). At high temperatures, especially with a catalyst, methane (CH₄) and water vapor react to give CO₂ and H₂:
CH₄ + 2H₂O → 4H₂ + 2CO₂
There are complications, and I’ve omitted a few steps. In particular, at the high temperatures (900C+) you need to steam-reform methane efficiently, hydrogen and CO₂ will react to produce water vapor and carbon monoxide:
H₂ + CO₂ → CO + H₂O
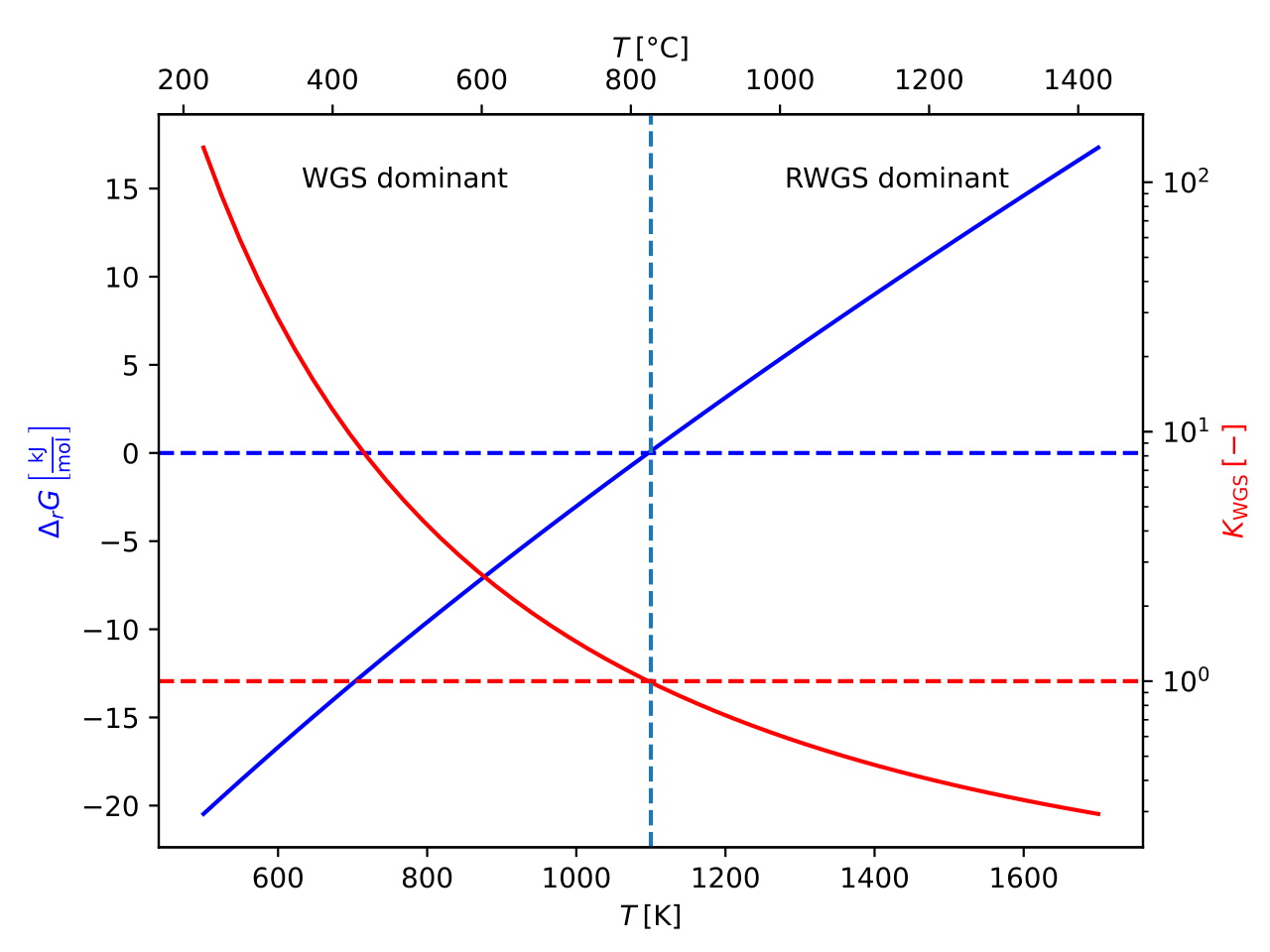
This is called the water-gas shift reaction. It reverses at lower temperatures to yield CO₂ and H₂ from CO and water vapor; the graph looks like this, courtesy of Wikipedia.
There are other issues with methane, most notably that it’s very difficult to store and transport. Like hydrogen, it requires special pressurized tanks.
However, steam reformation can be done with any number of hydrocarbons, which is why we’re going to use propane, C₃H₈. Propane has many virtues: it’s easy to pressurize and liquify, which is why you can buy a standard-sized 9-kilo/20-pound tank of it at Wal-Mart. It also starts undergoing steam reformation at much lower temperatures than methane: 300-350C rather than 900C or so, though it’s not very efficient at low temperatures.
Rakib et al. 2010 give a decent overview of steam reformation of propane. Their experiment was done at much lower temperatures than we’re shooting for, so even in the presence of a catalyst, yields were not that high (but neither was the rate of feedstock). They showed fairly conclusively that H₂ yields rise with temperature and the amount of steam used.
We’ll get into the details of doing steam reformation in a second, but I’ll first note that in an ideal world we’d just use pyrolysis (hat tip to Casey Handmer for the suggestion), which breaks C₃H₈ down directly with heat to give 4 molecules of H₂ and three atoms of black carbon, otherwise known as soot, with no carbon emissions entailed at all. It’s still very tempting to try this, for a few reasons:
a) One-pot reaction: heat it up and suck the hydrogen out; no messing with steam. No CO₂, either, if the carbon all gets deposited as soot1.
b) Low energy intensity compared to steam reformation. One kilo of hydrogen is about 500 moles, requiring 125 moles (5.51 kilos) of propane as feedstock. Propane has a specific heat of 1.68 kilojoules per kilo per Kelvin/degree Celsius, so heating it up from an ambient temperature of 20C to, say, 900C will require 1.478 megajoules per kilo of propane, or 8.14 megajoules per kilo of hydrogen thus produced. Assuming 50% electricity-to-useful-heat conversion efficiency, that’s about 4.5 kilowatt-hours—less than a buck’s worth of electricity.
c) Speed. This paper from Poland (written in the late ‘70s but mercifully in English) suggests propane breakdown happens fast, on the order of a second or so, though with lots of other hydrocarbons as byproducts. H₂ production more than quintupled (figure 2) when the temperature was beefed up from 991K (718C) to 1042K (769C). They didn’t go higher than this, though it’s probably safe to say that we’d see quite a bit of H₂ production at 900-odd Celsius, and that it would go quickly.
If I can get pyrolysis to work, then that’s what I’ll use. Empirical data at the 900-degree-plus Celsius range are sort of lacking for a question this basic, particularly without catalysts. (I’m not opposed to catalysts, but for a mass-manufactured backyard hydrogen generator, it needs to be cheap and available at the hardware store, or else you want to find a way to do without. I’ve seen references to iron oxide, i.e. plain rust; we’ll definitely be trying it.)
Why only the 900-degree range? Couldn’t we go higher? Pottery kilns go up to 1200C.
Hydrogen is notorious for seeping its way into almost everything and anything; it plays dirty with most steels, for example, as well as (at high temperatures) aluminum. This Department of Energy report gives a decent breakdown for the most common metals plus polymers; the best option, by far, is copper. But copper has a melting point of 1,085 C, so we’re going to need to stay comfortably below that temperature.
A potential alternative is ceramics that are fairly impervious to hydrogen. I know these exist and would let us go to higher temperatures, but I don’t know how much they cost.
Okay, so we’re going to use steam reforming for now.
That’s the assumption, although the first couple experiments will be with plain pyrolysis, because it’s the easiest to set up and would allow us to dispense with the water-shift reactor and just possibly the limewater (but probably not). Here is the chemical equation, just looking at the reactants and final products:
C₃H₈ + 6H₂O → 3CO₂ + 10H₂
(3CO₂ + 3Ca(OH) → 3CaCO₃ + 3H₂O)
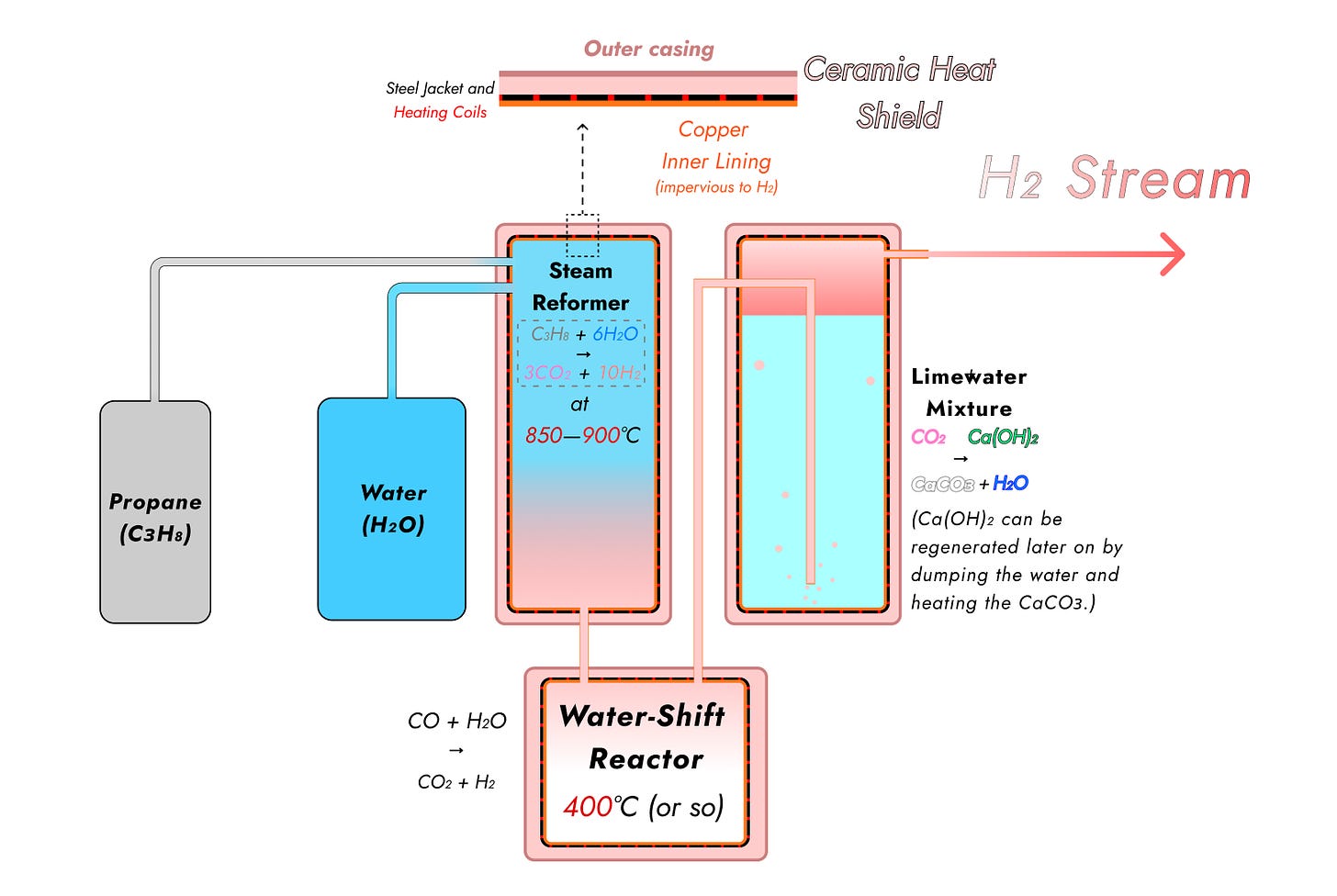
Here’s the schematic again.
For steam reformation, we’ll want at least six moles of steam for each mole of propane, although it’s plausible that we could cheap out on the steam a bit in the reformer proper and beef up the amount we use in the water-shift reactor, where it doesn’t need to be as hot. (Again, hard to find good empirical data). 6:1 is about 2.45 kilos of water for each kilo of propane; for a 20-pound/9-kilo propane tank, that means about 22 liters (remember, a kilo of water is a liter of water).
The water and propane go into the steam reformer, where they’re heated up to at least 850 centigrade and quite possibly higher, producing mostly CO₂ and hydrogen, along with the less-favorable pairing of more water and carbon monoxide. They are then yoinked into the water-shift reactor, which runs at a (relatively) cooler temperature of about 400 degrees Celsius (possibly even 350) and converts all but a handful of remaining CO molecules into CO₂.
The exhaust is then pumped into a whirling container of limewater. Limewater is a suspension of calcium hydroxide (Ca(OH)₂) in water; when CO₂ is bubbled into it, it reacts with the Ca(OH)₂ to give plain calcium carbonate, or chalk:
Ca(OH)₂ + CO₂ → CaCOɜ + H₂O
This reaction should occur faster with hot exhaust gases; we’ll also want the limewater swirling around a fair bit, and probably a second bubble-pump to circle the gas at the top of the chamber back into the limewater.
One kilo of hydrogen is about 500 moles: here’s a tweaked version of the relevant equations, ignoring the water-gas shift, first in moles:
50C₃H₈ + 300H₂O → 150CO₂ + 500H₂
150CO₂ + 150Ca(OH)₂ → 150CaCO₃ + 150H₂O
And now in kilograms:
2.2 kg C₃H₈ + 5.4kg H₂O → 6.6 kg CO₂ + 1 kg H₂
6.6 kg CO₂ + 11.11kg Ca(OH₂) → 15kg CaCO₂ + 2.7kg H₂O
Let’s talk energy costs for a bit.
Sure.
Let’s look at the energy costs for a full tank of propane, 9 kilos, which will require about 22 and a bit liters of water (let’s call it 23), produce a little over 4 kilos of hydrogen and about 27 kilos of CO₂. We’ll also need about 44 and a half kilos of Ca(OH)₂, which will react into about 60 kilos of calcium carbonate.
That’s a lot.
Agreed. We’ll only consider heating as everything else will be a rounding error.
Getting 23 liters of water from room temperature (20C) to steam at 900C occurs, as you doubtless recall from high-school chem class, in three stages. First, you’ve got to heat the water up to boiling point: the specific heat of water is 4.18 kilojoules per kilo/liter per degree centigrade. To get up to 100C, that’s 4.18 kJ * 23kg * 80K = 7.7 megajoules.
Thankfully, steam is easier to heat up: its specific heat is just 2 kilojoules per kilo per kelvin, so we need 2 kJ * 23kg * 800K = 36.8 megajoules, and—
You forgot the heat of vaporization.
…which, as I was about to say, is the biggest energy cost here: it’s a whopping 2,256 kilojoules per kilo, or 51.9 megajoules for 23 liters. Total energy cost for the water: 96.4 megajoules, or about 26.8 kilowatt-hours. At an average electricity rate of $0.15/kWh and 50% electricity-to-useful-heat conversion, that’s about eight bucks. Less if you’ve got a solar plant. You’re likely to want more steam than this for the water-shift reactor, but the point is it’s not going to break the bank.
The propane is easier to heat up, as I noted earlier: specific heat is 1.68 kJ/kg/K, and it’s already a gas once it’s out of the can, so a can of propane = 1.68 kJ * 9 kg * 880K = 13.3 megajoules, about 3.7 watt-hours. At 50% efficiency, this is $1-$1.20.
Last but not least, we need to deal with the limewater. At a density of 2.71g/cm³, the ~60 kilos of CaCO₃ needed per one-tank run occupy 22,140cm³, or a solid block about 28 centimeters on each side (a little bit less than a cubic foot).
To regenerate it into calcium oxide (which reacts with water to give Ca(OH)₂), we’ll have to heat it up—it starts decomposing somewhere in the upper 600s Celsius, but this gets much faster above 800C. We’ll shoot for 900C this time. (CaCO₃ is dirt-cheap and not very soluble in water, so you can always add more if you need to.) Because it’s not very soluble, we don’t need to boil the water from the limewater off—we’ll just check the pH to ensure it’s not so alkaline that it’s dangerous, then just pour it down the gutter.
Calcium carbonate has a specific heat of just 0.9 kilojoules per kilogram per Kelvin, so to get it up to 900C from 20C, we’ll need to spend 900 joules * 60kg * 880K = 47.5 megajoules, or around 13.2 kilowatt-hours. There’s also the enthalpy of reaction for actually decomposing it, which is 178 kilojoules per mole; the molar mass of CaCO₃ is about 100 grams exactly, so that’s 600 moles * 178 kJ = 106.8 megajoules, around 19.7 kilowatt-hours. Total cost at 50% electrical efficiency= 65.8 kilowatt-hours, or about ten dollars.
(Also, if you have some way to capture CO₂ around, that’s what’ll come out of the tailpipe when you’re done heating up the calcium carbonate. No emissions!)
Are there ways to save on energy?
(Note: this section takes the form of something of an internal dialog as I go back and forth rechecking numbers. But it’s often preferable, I’ve found, to see the author’s thought process…)
Well, you will get the enthalpy-of-decomposition energy back from breaking down the CaCO₃ when you Just Add Water to the calcium oxide, and there’s quite a bit of that there—65.3 kJ/mol, and we have 600 moles. 600 moles of water means about 10.81 liters; if you then add extra water, the 39.18 MJ (39,180 kJ) you get back should be enough to boil off (x*2,256kJ + (80K * 4.18kJ*x) liters; x = a little over 15 liters. That doesn’t account for additional energy loss and won’t get that steam up to the really high temperatures needed, but it’s not inconsiderable, because the heat of vaporization accounts for more than half the energy costs associated with the water. Equations:
CaCO₃ + heat → CaO + CO₂
CaO + H₂O → Ca(OH₂) + heat
We can also do things like coil the steam pipes around the gas-shift reactor and the throughput pipes. Remember, though, that this won’t allow us to make cuts to the caculated amounts of heat needed; it’ll only raise the efficiency of the energy we put in.
What about pressure?
This is probably the biggest issue: the pressures involved with heating gases up to the temperatures required are enormous well, fairly high. Their specific heats aren’t that high, though, and the reactions should go fast, which means that instead of trying to heat up everything at once (and blowing up the barn lab), we’ll want to use good sealed vacuums and ensure gas is handled in small increments.
Gas pressure can be calculated with just the number of moles, the heat involved, and the volume of the space in question. Let’s assume our steam reformer can be safely operated at 20psi2 and comprises a cylinder that’s 150cm tall and has a radius of 30cm (so about 4.24m³). Using our trusty gas-law calculator, what we’ll find is that we’re allowed just under 60 moles of gas at a time at 900C. (But see next below for ideal boiler geometry, which I didn’t use here).
Note that because it’s moles, not mass, that matters with pressure, the litmus test isn’t how much propane and steam it can safely handle, but how much hydrogen and CO₂, since 7 moles (1 propane + 6 water) enter and 13 (3 CO₂ and 10 H₂) leave. This means we’re allowed 4.61 moles (203g) of propane and 27.7 moles (504g) of water at a time: about 45 runs per tank of propane.
N.B. that the lower temperature of the water-shift reactor means it can be considerably smaller. 60 moles of gas at 400C in a chamber operated at 20psi will have a volume of about 2.44 m³. To minimize the surface area for a given volume, you want a cylinder whose height is equal to the diameter of the base, in which case 2r = h and the volume reduces to (πh³)/4. An ideal boiler with a volume of 2.44m³ then has a height and diameter of about 1.46 meters, vs. about 1.75m for the ideal 4.24m³-boiler.
And the energy costs of heating up the copper and steel…?
These, it turns out, are surprisingly pricy!
Copper’s specific heat is very low, and steel’s isn’t much higher. Steel’s (well, iron’s) is 420 joules per kilo per kelvin. We’ll work with steel.
For a boiler whose height and diameter are ~1.75m each, the radius is ~87.5cm; surface area is about 14.43m². If we beef this up to 14.6m² to make the math easier and account for that figure being inner surface area, then if we assume a two-centimeter thick wall of steel, we have about 0.29m³ of steel. At 8 (metric) tons/m³, that’s about 2,320 kilos of steel; 420 J * 880K * 2,320 kg = …
…857 megajoules, or 476 kWh at half efficiency = $71 of electricity. I’m pretty sure I haven’t messed up any major numbers, but let’s see.
At least once you’ve gotten it that hot, you just need to maintain it.
Well, I suspect there are a few ways around this. The resistance heater can be placed inside the steam reformer rather than running around the walls. The copper surface might well include the steam pipes feeding the boiler.
Also, 20psi might be far too cautious. The physics of when a pressure vessel can vs. can’t be trusted to explode are complex, but here’s a handout (uploaded to Weebly by an enterprising cheating enthusiast) by an engineering prof giving us the example (problem 4) of
Calculate the minimum wall thickness for a cylindrical vessel that is to carry a gas at a pressure of 1400 psi. The diameter of the vessel is 2 ft, and the stress is limited to 12 ksi.
The minimum wall size in this example is 1.4 inches. But we can take the equation and play around with it, assuming that the problem example is about the sort of thing you’d see in the real world and doesn’t involve walls made of either unobtanium or paper.
Allowed tangential stress σ = (p·D)/(2t), where t is the the wall thickness, p is the pressure rating, and D is the inner diameter (in whatever units you’re using for pressure and diameter). The inner diameter of our cylinder is 1.75m = 69 inches; ksi is kilopounds per square inch, so if 12,000 psi is the allowed stress rating for the sort of vessel we need, (for which I have no intuition), then we could beef up our internal pressure to 200 psi with wall thickness of no more than about 1.74 inches, or about 4.4 cm.
If we do a bit of algebra, what we’ll see is that pressure rating p = (2·σ·t)/(D), or in other words that all else being equal, your allowable pressure will double if you halve the diameter. Alternatively, t = (p·D)/(2·σ), so halving the diameter also allows you to cut the the wall thickness by two.
The unknown here is σ, which won’t be entirely independent of the other parameters, but this is an informative model. The ideal, one presumes, is a tall, skinny cylinder; there’s an interesting math problem to be done here of the form Given a constant pressure, volume and stress rating, what are the radius and height of a cylinder that minimizes the amount of material used in its walls?, but we’ll deal with it once we have actual quotes from manufacturers in hand. The heating element is probably most ideally inside the tube, but the joints where it and its wiring contact the outside world might be weak spots. The walls will need to be wrapped in a very good insulator/heat reflector (hence the ceramics). You might want to bury it in a hole in the ground, both for insulation and so that consequences are minimized if things, for whatever reason, go Badly. (I will try my damnedest to make sure the reactors are really presssure-tolerant).
At 200psi and 900 degrees Celsius, 60 moles of gas would only require a volume of 0.424m³. If we have a cylinder where the height is five times the diameter, then the radius r is 42,400 cm³ = πr²(10r) = about 11.05 cm, and the height is a little over a meter. Surface area is (2πr(10r + r) or 22πr²) = about 8,440cm²; if we round up to 8600 (0.86m²) to account for the inner vs. outer surface area, a 2.5-cm-inch-thick wall of steel means somewhere in the neighborhood of about 0.0215m³ of material, weighing 172 kilos. 420J * 880K * 172 = ~63.6 MJ, or about 35 kWh at 50% efficiency—a little over five bucks. We’ll need less than that (420J * 380K * 172 = 27.5 MJ → 15.3 kWh = $2.30) for the water-gas reactor.
It seems quite clear that the way to defeat the energy-cost problem here is to make sure the steam reformer, in particular, has a pretty high pressure rating.
Gimme the total opex costs here.
For 4 kilos of hydrogen: a $25 refill for a propane tank, 40ish liters of (tap?) water (11 for the limewater and then 23-and-then-some for the reactors) for maybe a nickel. Cost of heating the water: $8. Cost of heating the propane: $1.20. Cost of heating and regenerating the CaCOɜ: $10. Cost of keeping the reactors hot: maybe $7.50, if we assume 200-psi reactor chambers. Total cost is about $50, so ~$12.50 per kilo of hydrogen.
It’s not cheap exactly but many places have electricity that’s cheaper than $0.15 per kilowatt-hour. If you have $0.10/kWh electricity, you’re looking at aroung $8 a kilo. From what I can tell, though, propane refill costs don’t scale much (please leave a comment if I’m wrong on this).
And the capex costs? You were thinking of selling these, right?
Remains to be seen. “Pressure vessel that can handle mid-hundreds PSI, the interior has to be totally copper” is a custom job.
(Edit, 8/11/23: I am happy to announce that the main reactions can take place in LARGE QUARTZ TUBE. See discussion towards the end of the (first?) update post.)
What is the carbon footprint of running this?
Total electricity cost per run is ~175 kWh. You’ll emit about 27 kilos of CO₂ (from the carbon in the propane) when you regenerate the calcium carbonate to make calcium oxide (which you can sequester if you’d like—comes out in a pure stream from the exhaust valve). Average carbon intensity of electricity in the US right now is around 389 grams of CO₂ per kWh, so about 68 more kilos. Less if you’re on solar. Total footprint = 95 kilos for a full run, or around 24 kilos of CO₂ per kilo of hydrogen.
Compare the expensive generator on Amazon: 28 kilos of CO₂ emitted per kilo of hydrogen at prevailing electricity intensities. And you’d have to run it for two months straight to get as much as one of these should be able to produce in an afternoon.
I am not convinced this is safe.
Don’t be an imbecile around hydrogen or things running at 900 degrees centigrade.
And if you’re not confident about the safety? Don’t buy one.
Though I will say that one of the first things I’ll be doing with the Kickstarter proceeds is getting some very good legal advice.
I would like to support this venture.
I am holding off on starting the Kickstarter until I have a better sense of the costs. I have requests for quotes out to a couple of outfits that manufacture good pressure vessels.
I can, however, promise fantastic bumper stickers for backers.
If you can support this venture by providing an abandoned barn within a two-hour drive of DC, please get in touch.
How do I do that?
DM me on Twitter or get in touch with me by email.
(Update 1, Update 2, Update 3).
I suspect this is unlikely, so the limewater reactor will probably have to stay.
But see discussion later on—this pressure rating will be way too low.





Why do you have to use high pressure? You seem to be assuming you fill the reaction vessel with cold gas, seal it, and heat it up. Why can’t you just run it continuously with pipes flowing into and out of the reaction vessel, at atmospheric pressure or barely above? I looked through your essay but didn’t find a justification for the high pressure. Maybe I’m being dumb.
Also, you can recover a lot of the process heat with a countercurrent heat exchanger on the input and output pipes.