Manufacturing a Backyard Hydrogen Generator: Update 3
A semi-final schematic, with a name, before I set up a Kickstarter
Continued from here. Recap of all four posts is here.
As we saw last time, propane is an expensive way to get your hydrogen. What if you used methane?
If you have a gas line, then…why not?. Just make sure you have a really good tradesman on speed dial. I do. Then you’ve got a much cheaper feedstock.
Utility natural gas is almost all methane, and is sold (in the US) by the “therm”, which is 100 cubic feet. As of the moment (on the East Coast), municipal rates per therm are about 35 cents; they can shoot up as high as a dollar during energy crunches like the summer of 2022. A cubic foot of methane is 1.195 moles, so one therm will buy you about 1.96 kilos. Far cheaper than propane, though you do need to have a gas line that you’re willing to competently tinker with.
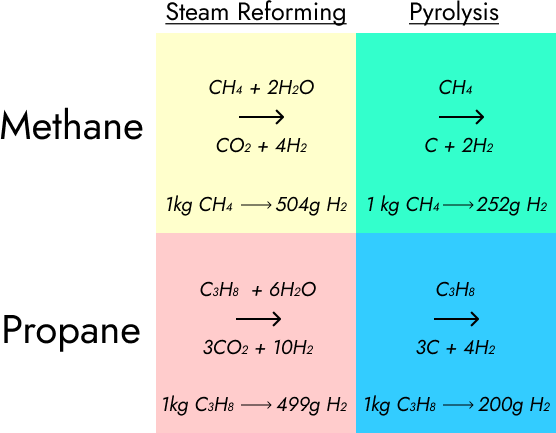
CH₄ is a cheap feedstock, while C₃H₈ is a pricey one. Reforming is cheap but dirty, while pyrolysis is pricey, but clean, and should also require somewhat less equipment. The ideal for Pythia (tentatively named after the Oracle of Delphi, whose hallucinations may have been caused by natural gas seeping from the ground) is that it should be able to do any of these.
Let’s take another look at the equations, inputs and outputs.
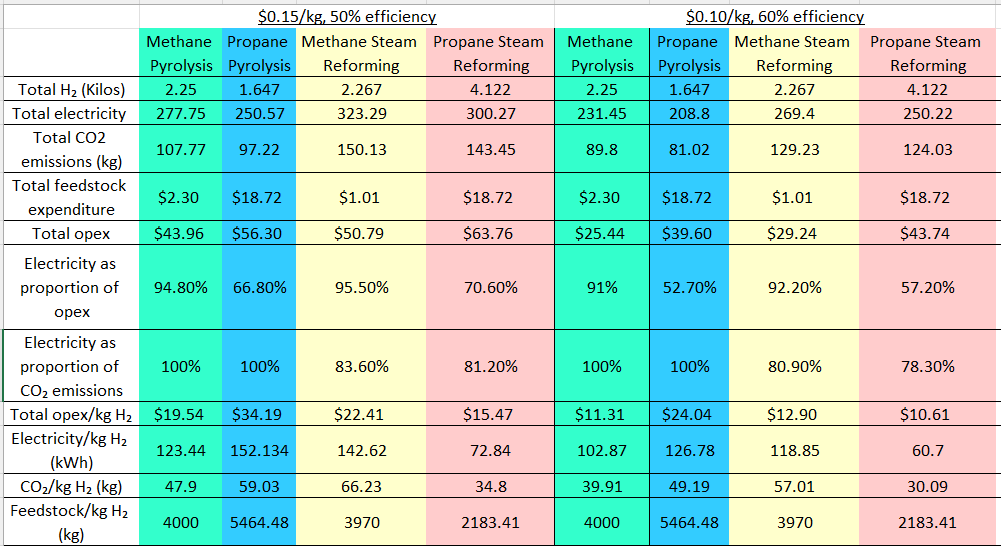
Let’s run the Python script and see what we get. The following spreadsheet assumes that we’ve got 9 kilos of feedstock, that methane costs 50 cents a therm and propane $4 a gallon, that our electricity source has a carbon intensity of 389 grams per kWh, and that electricity is the only cause of carbon emissions with pyrolysis.
Propane pyrolysis is out because it’s below the Pareto frontier on pretty much every metric—carbon intensity, cost of feedstock and electricity consumption. It may make sense if you have access to cheap, low-carbon electricity but have no natural gas line.
Steam reforming is not as bad as feared. Unless your electricity mix is overwhelmingly carbon-neutral, most of the carbon emissions from the process will be from electricity consumption rather than direct CO₂ emissions out of the tank.
Every other option is a potential Pareto optimization. With extremely pricey electricity or (more likelY) no access to a natural gas line, propane steam reforming is sensible. Methane pyrolysis makes some sense if you have low-carbon electricity. If you don’t have low-carbon electricity (or are OK with some carbon emissions from the process) and have a natural gas hookup, methane steam reforming is what it makes sense to do. Methane pyrolysis, however, is hard, for reasons we’ll see later.
What about the rest of it?
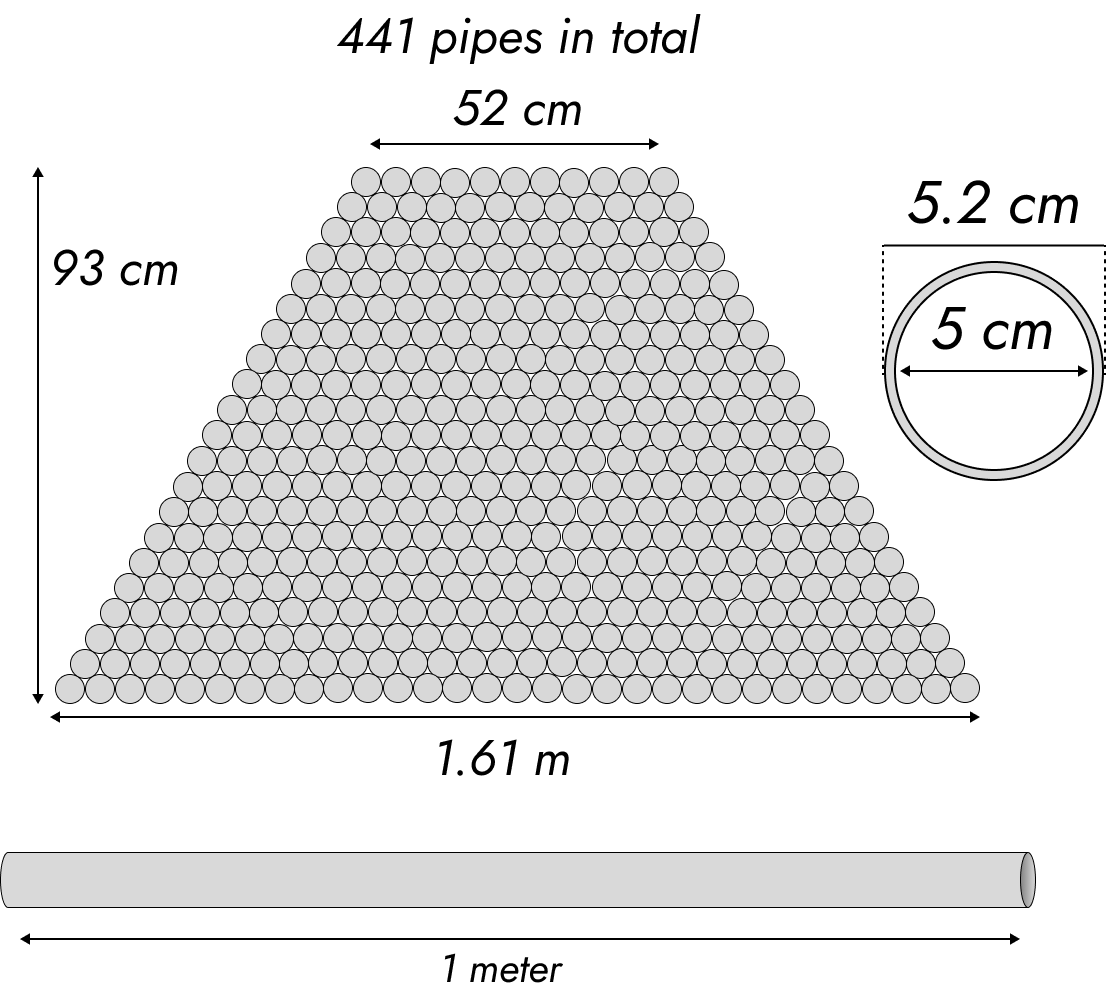
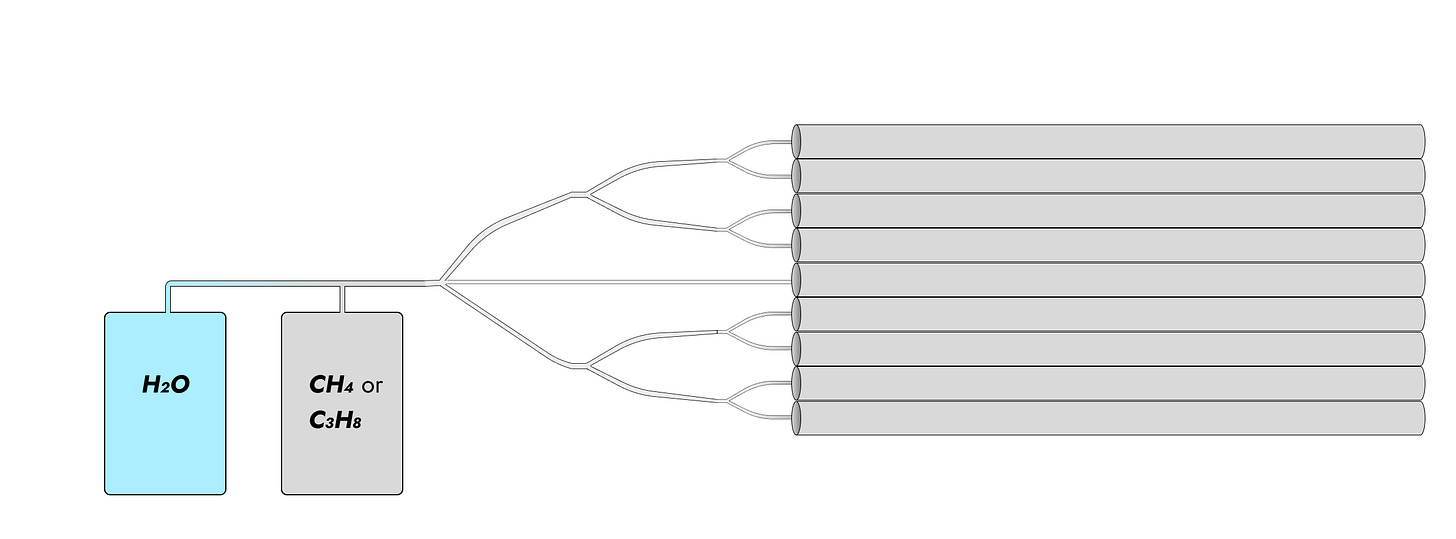
As we saw in Update 2, we can get good results from using a pile of pipes. Here’s a possible setup.
Catalysts have to be chosen carefully here. Nickel does poorly with methane pyrolysis at high temperatures. Iron works fairly well, however. These researchers had 45m² of iron catalyst surface in a reactor at about 1 bar of pressure and a four-second residence time, with no indication of deactivation, though they had to go up to 1100C. Iron does catalyze steam reforming, but it doesn’t do so anywhere near as well as nickel.
There are probably a number of options here if we want to make both steam reforming and pyrolysis possible in the same device. Note that constantan’s solidus point is only 1083 C. We can find another alloy, particularly if pyrolysis of propane isn’t a priority, and fairly secure in the knowledge that what works for methane will work for propane as well. Steam reforming is generally easier than pyrolysis, so my inclination is to make the outer pipes out of iron, and the feeder pipes out of nickel—particularly since the feeder pipes will have to deal with higher presures.
A cross-section of pipe should look like this.
The holes in the feeder can be opened and closed (there’s probably a sliding element). Gas (feedstock with or without steam) enters the feeder and is heated up to a screamingly high temperature by the heating element. Hydrogen escapes. If pyrolysis is used, the high speeds of the gas should be sufficient to knock soot off the walls and onto the floor of the pipe.
It took me forever to try and draw the valve branching here, so we’ll use a more abstract schematic. There are two more chambers to deal with at this point.
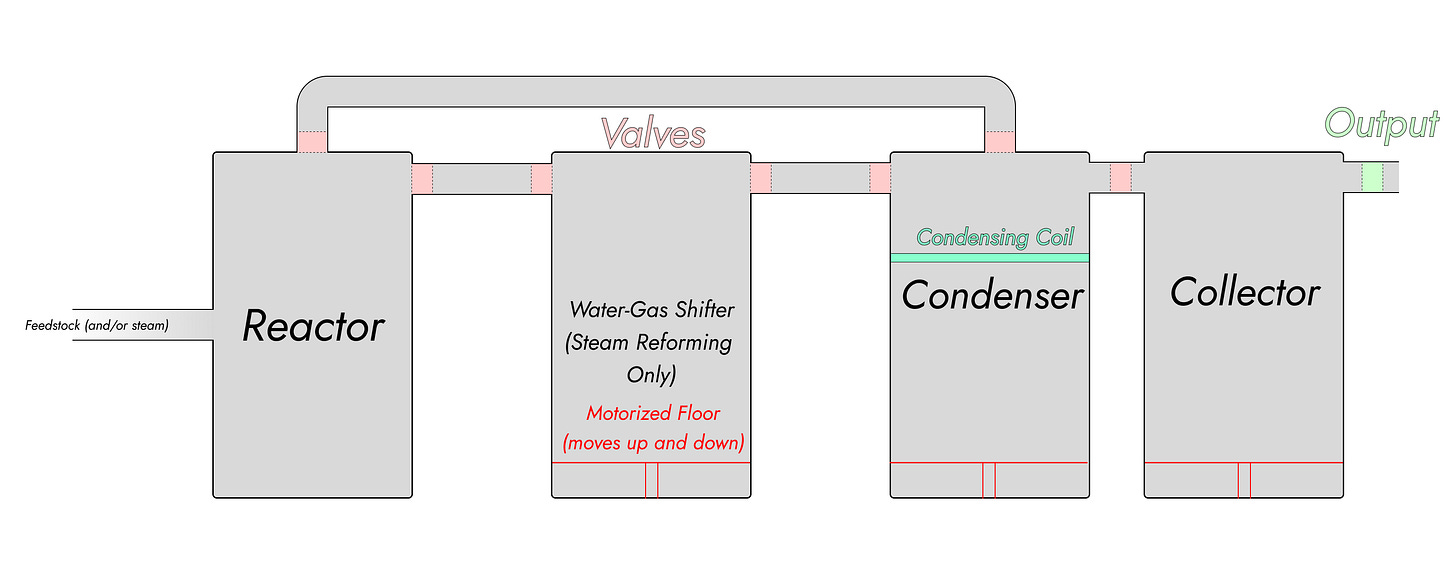
Pyrolysis doesn’t need to deal with the water-gas shift, so that reactor can be skipped. Hydrogen needs to be persuaded to move out from the pipes into a lower-pressure, larger area; that’s the water-gas shifter followed by the condenser. Once a decent amount of gas has condensed, it’s off to the collector—through limewater if we’re using steam reforming. (Did I calculate the energy cost of lime regeneration for that Excel spreadsheet? Admittedly, no, but we dealt with it in the first post: at 15 cents a kilowatt-hour, it’s about $2.50 per kilo of H₂).
This all looks Big, of course. The biggest remaining issue is probably the sheer size of the collector: 4 kilos of hydrogen is 1,980 moles, and at standard temperature and pressure that’s 48 cubic meters. If you’re inflating weather balloons, that’s probably fine. If you’re doing chemistry experiments, we’ll presume you probably don’t need 4 kilos at once, and that if you do you’ll use a weather balloon to store it in. So perhaps the collecter can be dispensed with except for the limewater, which will also act as the condenser.
That’s pretty much it for now.
Wait, what? Yeah. The really big issues have been worked out. We know that backyard hydrogen production, certainly via steam regeneration and probably via pyrolysis, is feasible with cheap materials, with relatively low opex costs. And I don’t need to be competitive with industrial wholesale prices; I just need to be competitive with the welding supply store.
Before I set up a true Kickstarter, I would really like to get a consultation on product liability1. If you’re interested in supporting this venture pre-Kickstarter, I have set up a paid tier. Product development updates will be at least half paid tier.
If this turns out to have too many liability snags, did you know that the main player in olivine weathering is still, fatally, using sand?



What do you intend to do about the residual CO? Water gas shift does not go to completion, especially if you're using a HTS without catalyst.