(Note: At the time of publishing I am working on Update 3, in which it becomes clear that steam reforming remains a viable option much of the time, at least looking at energy costs and CO₂ emissions. Methane pyrolysis is something of a Hard Problem, but should be feasible).
A few hours after writing Update 1 it finally occurred to me that pyrolyzing propane in a chamber is going to leave soot all over the walls, which you’ll have to clean out. No more sealed quartz jars that you never open again! Oops.
What are our other options?
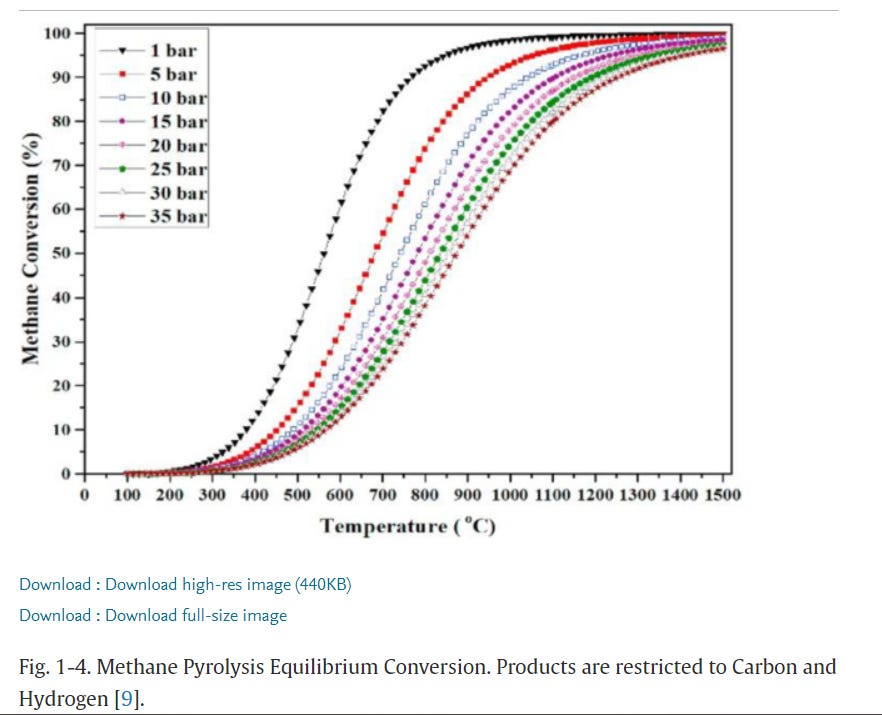
Pyrolysis of propane is a complicated problem. In general, the equilibrium ratio of H₂ to hydrocarbons rises with temperature, but decreases with pressure. Here’s methane pyrolysis:
But the rate of pyrolysis increases with pressure. In other words, suppose it takes t seconds at 700C and 1 bar of pressure (about 14.5 psi) to hit the equilibrium ratio (somewhere around 85% of all CH₄ pyrolized.) If you then double the pressure to 2 bars and run the process again, you’ll hit a lower equilibrium (eyeballing it,maybe 65% of all methane pyrolized), but do so faster. To get to the same equilibrium ratio you’ll need to raise the temperature.
In a way, this makes things easier: scrap the high pressures. Methane pyrolysis is relatively easy to model because methane has only one carbon; propane has three, which means the intermediates are other hydrocarbons, including methane.
Switching all that carbon to soot is a complex one. Successful pyrolysis will break down each mole of propane into three moles of atomic carbon (C), which won’t affect the gas pressure in the chamber, and four moles of H₂, which will—remember that it’s number of moles, not amount of mass, that affects pressure, so we’ll be effectively quadrupling the internal pressure.
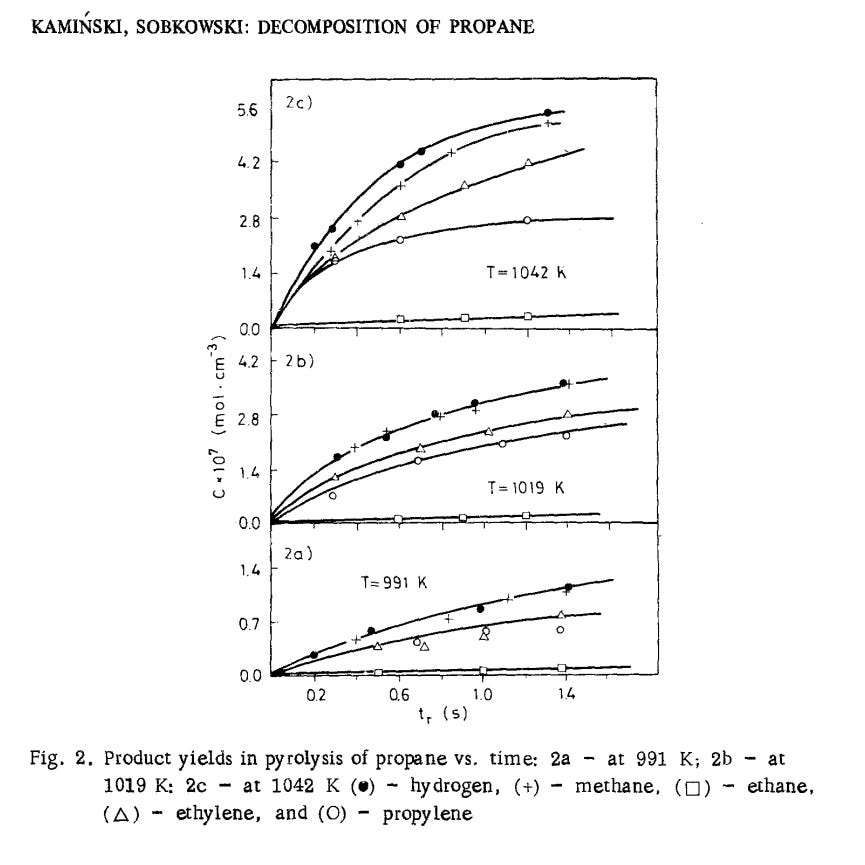
As we’ve seen, pyrolysis happens pretty fast:
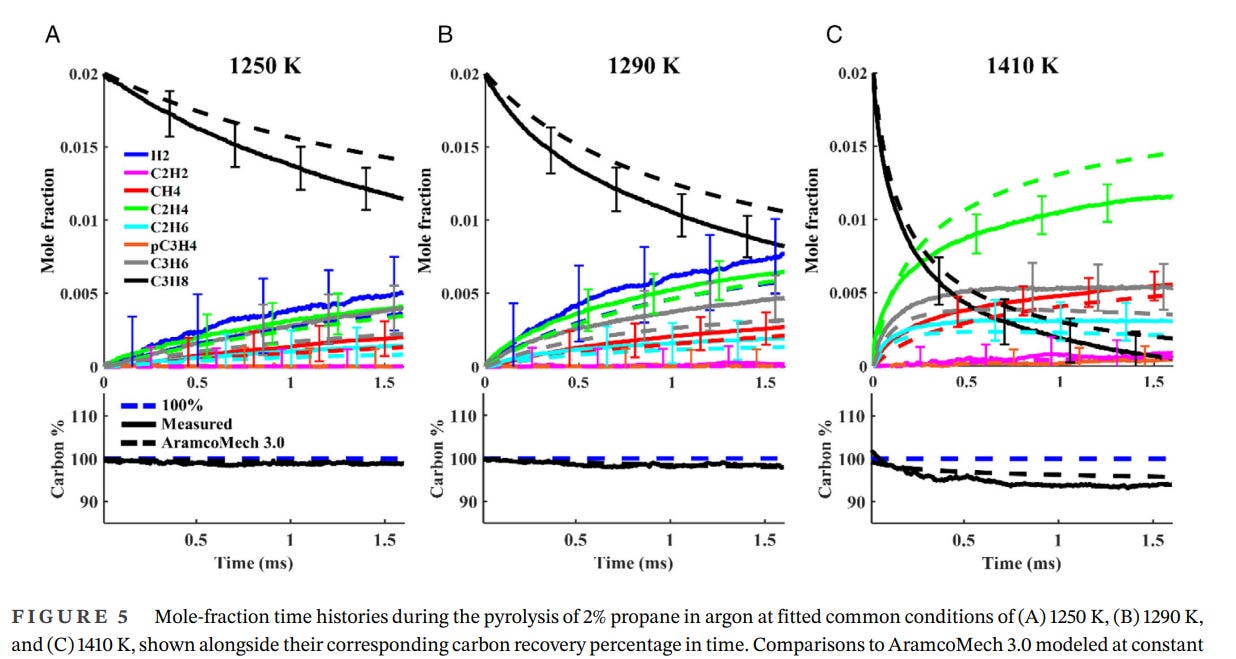
…however, there are some indications that there’s a limit on effective upper temperature. This article has a relevant chart:
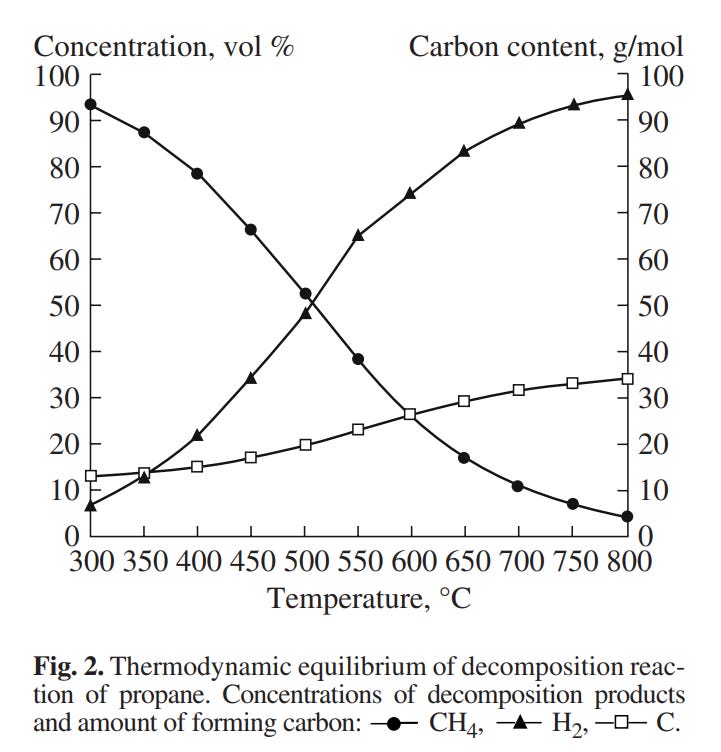
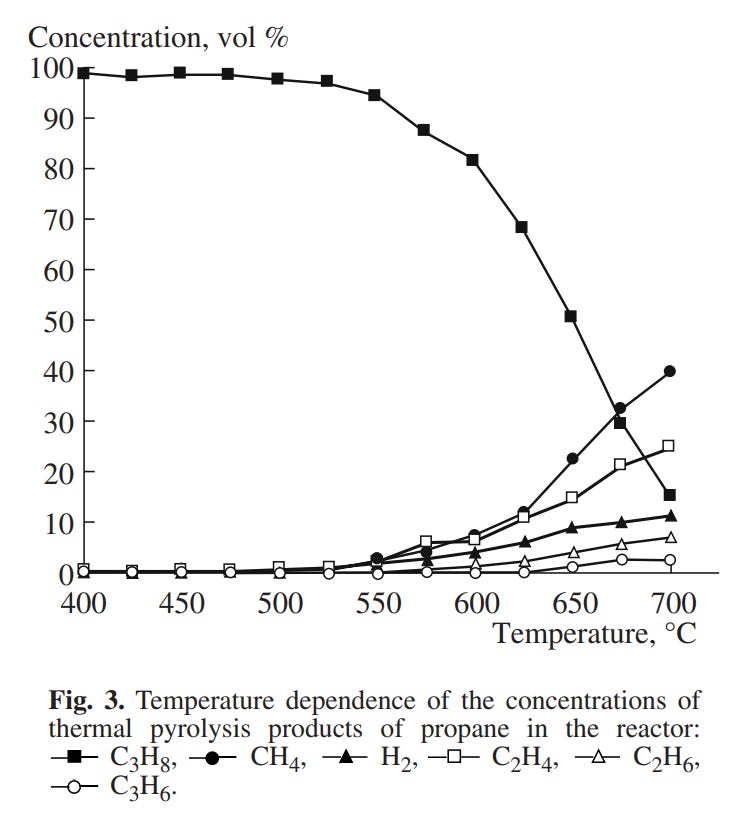
Another relevant paper is this one (Solov’ëv et al.), which also concerns itself with propane specifically. Note particular Figure 3:
That’s just the equilibrium between hydrogen and methane at atmospheric pressure (~1 bar, 14.5 psi), though, not the actual results of their propane decomposition, which look like this:
Solov’ëv et al. were mainly attempting to measure the effect on catalysts, so for residence time, we have to work backwards from their experimental setup.
Their reactor was small—an inner diameter of just 24mm, and a height of 150mm—giving a volume of about 68,000 mm³, or about 68 cm³. Their flow rate was 1 liter per hour (presumably at STP, where one mole of gas takes up 22.4 liters); that gives a residence time of about 4 minutes, about 0.003037 moles of gas in the chamber at any given point, and an internal pressure of about 63.2 psi.
Could we do better?
That’s a tiny tube. It’s sensible for doing experiments, of course, but not for backyard processes (I hesitate to say industrial processes because if we already have good ways of producing industrial-scale amounts of hydrogen).
Recall that raising temperature and pressure will both increase the reaction rate of methane pyrolysis, but that the equilibrium amount of hydrogen will be higher as pressure drops, so there are inherent tradeoffs here—recall that Kamiński and Sobkowski used pretty low pressures, while Solov’ëv et al. used high ones, so their results aren’t directly comparable.
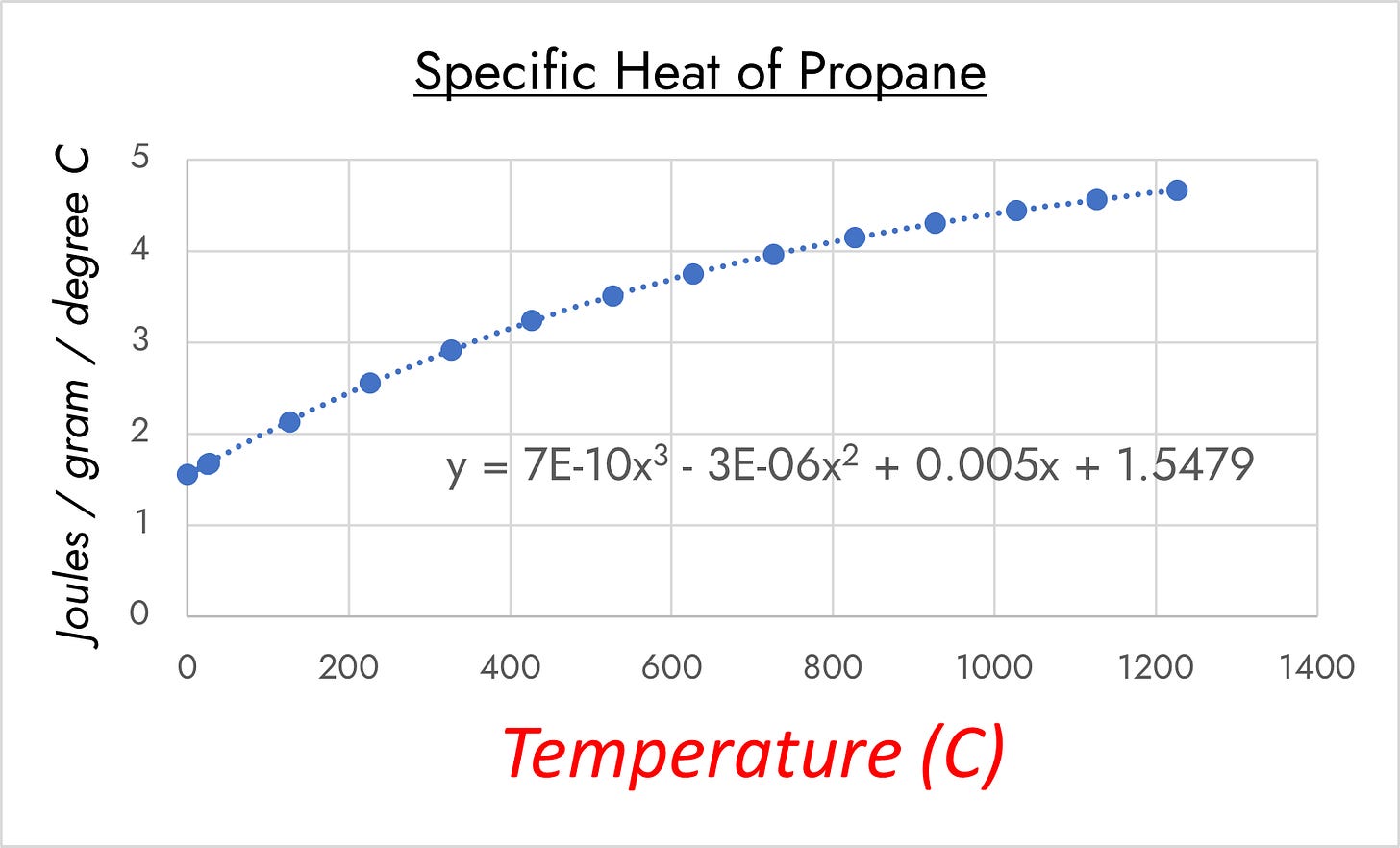
Time to call in the Python scripts and more math. The specific heat of propane varies over the span we’re looking at (source):
Integrating from 25C1 to 900C gives a value of about 130.15 kilojoules per gram2; compare the standard enthalpy of formation of propane at 25C, which is about 50.11 kilojoules per gram3. This Python script takes various parameters (which can be default values or inputted, as you please) and spits out calculated values of interest, so we’ll use it.
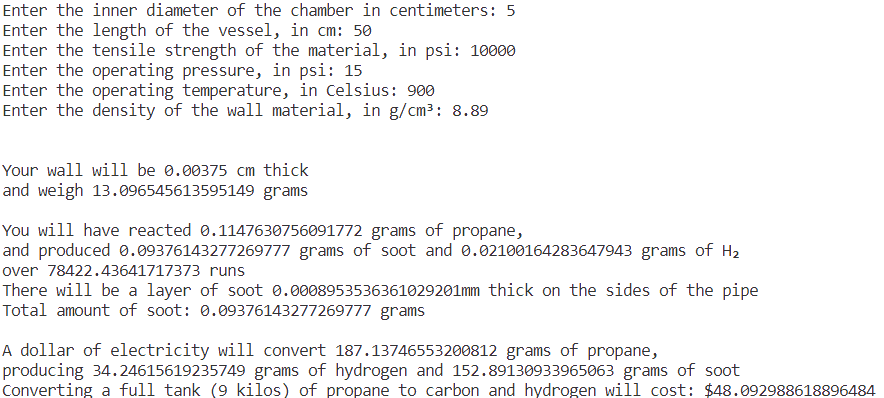
Let’s start thinking about reaction chambers again. We don’t want to use super-fancy supported catalysts because they’d have to be replenished, so let’s just make our pipes out of a halfway decent catalyst and hope it gives a boost to pyrolysis. (Remember, pyrolysis happens fast.) We’ll use a copper-nickel alloy in a 5:4 ratio by mass for now, from this paper, otherwise known as Constantan.

Constantan’s solidus point is 1083C, so the mid-900s C should be OK. The density is about 8.89 g/cm³. Tensile strength is hard to figure out: Wikipedia cites a book on copper alloys that only gives a range of 455-860 MPa (about 65,000-125,000 psi), with no reference to temperature. We can try and compare Nickel 201, which is almost all nickel with a few other metals, and whose tensile strength is lower than the whole range given for constantan: tensile strength drops from about 58,500 psi at 20C to 22,200 psi at 649C. Let’s assume a tensile strength of 10,000 psi (10 ksi) for constantan at 900C for a first pass.
Because the molecules in pyrolysis are bouncing off the walls of the chamber tens of thousands of times a second, soot deposition shouldn’t be a problem with larger chambers. Let’s go for the following parameters:
pressure is 15 psi (about 1 bar), operated at 900C.
the tube is 5 centimeters in inner diameter and 50 centimeters long.
the material parameters are those of constantan: 8.89 g/cm³ and tensile strength of about 10,000.
God that electricity cost is obscene. And seventy-eight thousand runs? And how are you going to get the soot out?
You’d rather go to the welding store and pay a few hundred a kilo? Well, no, but— Always make pessimistic assumptions, then you’ve got some breathing room. Who said we were wedded to a single pipe?
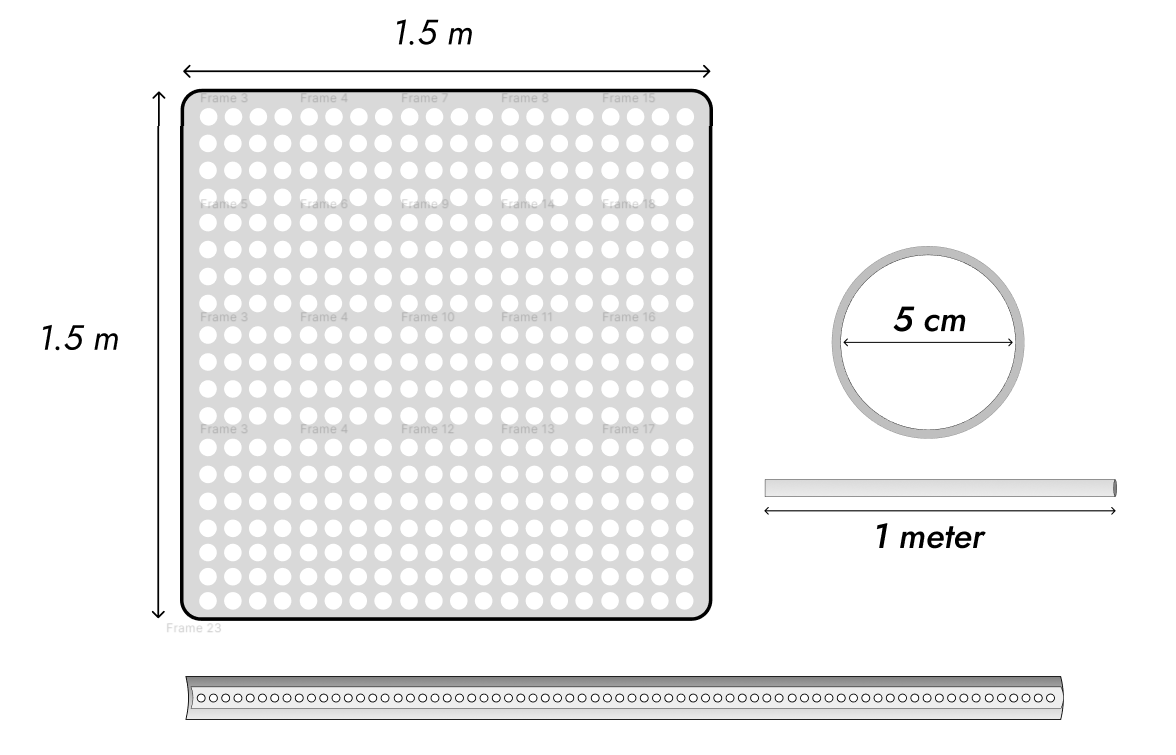
Four hundred pipes? It’s actually 380, I’m only so good at making diagrams. Please pretend there are 400 pipes. So, a 2.25m³ block, with about 0.785m³ hollowed out for the pipes, leaving about 1.46m³ for the metal. Copper is—this will cost six figures. Fine, I’ll actually pay attention to the wall thickness, which the program said would only have to be 0.00375cm. I ignored the wall thickness because I can’t draw circles that small. 0.05cm, please. I’m paranoid. Fine. I’m not drawing this again.
We’ve now got a packing problem with pipes 5.1 cm in diameter. We’ll try doing 500 of them. That’s only, wait, 793cm³ of constantan. A bit over 7 kilos. That’s basically foil. Good, that means we can ignore the effect of pressure on the wall thickness. Even centimeter-thick walls would only be about 18,849cm³ of metal, about 168 kilos. Nickel is twenty bucks a kilo right now, copper’s about eight, that’s about $13 a kilo. A little over two grand.
Okay, now what? The 50cm-long pipes converted about 0.1145 grams of propane per run, so the meter-long ones will convert about 0.229. 39,000 runs to convert a tank of propane. No, 78 runs. We have 500 pipes. You remember that pyrolysis at high temps takes only a few seconds, right?
That thingy in the middle of the pipe is the feeder pipe; we’ll put a resistance heater in the center, probably some silicon carbide, and feed propane through it. Silicon carbide costs only a few bucks a kilo, gets crazy hot, and most of its heat will go to the gas.
Soot cleaning. Yeah, I was about to get to that. We’ve got 1963 cm² of area in each pipe, a little over 980,000 cm² in the whole thing. 7,350 grams of soot total in a 25-gallon tank, about 11,000 cm³. That’s about a tenth of a millimeter.
Is that going to be enough to kill the catalyst early on? We don’t know, but I kinda doubt it. Solov’ëv et al. had 0.01 grams of catalyst in each of their experiments, were running 1.6 grams of propane over it per hour, and didn’t have problems with the catalyst for hours.
They were using nanoparticles, probably higher surface area. Fine, let’s take a look at the paper they cited. Check figure 5.
No serious hit to performance in Solov’ëv et al. for about 3 hours, by which point they’d pumped 4.8 grams of propane in. 0.01g of catalyst means 1.5m² of total surface area; if we’re inclined to invent a unit to compare, that’s 3.2 g/m² before the catalyst started having problems. We’re looking at 9,000 grams of propane over…aw shucks 98m². Our ratio is about 30 times higher. Hey, at least it ain’t 3000 times higher.
My assumption is that a fine layer of soot is the main culprit here. We’ll get 7.35 kilos over the course of the run, 14.7 grams per pipe, occupying 9.8cm³ in a volume of about 1963cm³. That’s about half a percentage point of the total area, with near-negligible effect on the geometry.
The thing about a mostly-smooth pipe interior is that you can wiggle it a bit and things should fall off, because the catalyst isn’t made of nanoparticles with nooks and crannies all over the place. There’s a possibility here that this won’t work (electrostatic forces?). We might also get lucky and have the propane do the work for us. Suppose the inner tube is only a couple centimeters in diameter, so that the area is 4% of the pipe and pressure is 25 times higher—about 362.5 psi—That sounds unworkable. Fine, we’ll handwave it and pretend it’s 50 psi. We’ve flushed out enough hydrogen that pressure in the pipe is only 1 psi. We’ll use this calculator, pretend the propane is already at 300 degrees Celsius, and choose an orifice size at random. (Grotesque.) Through an opening of 0.03 inches (the largest they have), we’re looking at a flow rate of about 4900 standard cubic centimeters, or five liters, per minute per an opening of about 0.02cm² at 300 Celsius.
The math gets difficult here, but I suspect the numbers allow us quite a bit of wiggle room. A cylinder of 4900 cubic centimeters, of which the bases are 0.02cm², is about 2.45 kilometers long. If you’re travelling that rate in a minute you’re going about 90 miles an hour, which should be well more than fast enough to knock a bunch of soot off the walls. We can also increase the speed by alternating which holes of the inner tube are open on any given run.
Is that it?
No, not entirely. We still need a fairly capacious condenser to get the hydrogen to migrate out of the chamber—calculations on that to come—and eventually you will have to get the carbon black out of the pipe. Tilting the pipe and giving it a propane flush into a collection chamber, with the propane going back to be pyrolized, is probably feasible. And there might still be complicated problems to solve with pyrolysis, because there are so many intermediaries. It’s also fairly expensive.
But I think the really hard problem is over.
Continue here.
We’ll have to assume a nice summer afternoon to square this value with the enthalpy of formation.
God I hope I haven’t misplaced a decimal place.
Found by taking an average value of -2210 kJ/mol from the NIST data, then dividing by the molar mass.